Stochastic Non-Equilibrium Thermodyamics
|
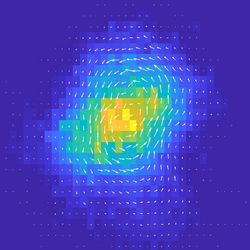
We are interested in measuring a distance from equilibrium, and relating this to changes in material phase, and to the function of biological systems. Ben Machta (Yale Physics) develops novel frameworks for estimating the irreversibility of non-equilibrium systems which have proven powerful for understanding transitions in non-linear systems. Thus, we apply these frameworks to simplied and controllable ‘active’ experimental systems that exhibit stochastic dynamics. We also study these systems in simulations of reaction-diffusion dynamics. Publications: [Nat Comms, 2018][Adv Func Mat, 2020],[Nat Comms, 2021] People: Daniel S. Seara, Vikrant Yadav |
 |
Experimental Biological De Novo Assembly
|
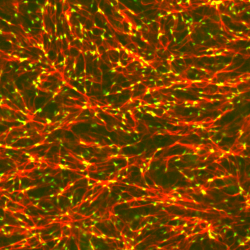
The non-equilibrium thermodynamics of biological assembly addresses how living systems build and maintain complex structures—such as cytoskeletal networks, membrane domains, and organelles—by continuously consuming energy. Unlike equilibrium self-assembly, which is driven by the minimization of free energy and leads to static, reversible structures, biological assembly occurs in open systems maintained far from equilibrium. Energy, often in the form of ATP hydrolysis, fuels active processes like filament polymerization, motor-driven transport, and enzymatic modification, which bias assembly pathways and sustain dynamic steady states. This energy input allows cells to generate gradients, rectify fluctuations, and break detailed balance, enabling the formation of structures with spatial organization, temporal dynamics, and mechanical function unattainable in passive systems. Understanding these assemblies requires tracking not only the organization of matter, but also the continuous fluxes of energy and entropy production that maintain them. (right) Remodeling of an F-actin network (red) by myosin II (green) |
 |
Active Wetting of Living Cells & Tissues
| Active wetting refers to the spreading and dewetting behaviors of active materials—such as cells or tissues —on solid or fluid substrates. Unlike passive droplets, whose wetting dynamics are governed by equilibrium surface energies and interfacial tensions, active systems continually consume energy, giving rise to non-equilibrium stresses and fluxes that modify wetting behavior. This energy input can generate effective surface tensions, induce spontaneous flows, or break symmetries in contact line dynamics. Understanding the energetic properties of active wetting thus requires going beyond classical Young–Laplace theory to incorporate energy dissipation, active stress generation, and feedback between shape changes and internal dynamics. Such systems often exhibit novel behaviors—like spontaneous spreading, contractile dewetting, or shape oscillations—that reflect the continuous conversion of chemical energy into mechanical work at interfaces.
Your browser does not support the video tag.
|